
What fruit flies are teaching us about the need for sleep
From fruit flies to flamingos, every animal with a nervous system needs sleep. But exactly why isn’t understood. Scientists do know that sleep is regulated by two parallel mechanisms: the circadian clock and sleep homeostasis. The longer we stay awake, the stronger the homeostatic pressure to sleep becomes. But what are the precise signals of this need inside the brain, and how does that signal translate into the neural activity that drives rest?
Recent studies using fruit flies (Drosophila), undertaken by University of Oxford researchers Dr Raffaele Sarnataro & Dr Peter S. Hasenhuetl, are providing new insights into how sleep is controlled. Their studies have found that neurons in a key sleep-regulating region of the fly brain, the dorsal fan-shaped body (dFBNs), track sleep need via the energy flowing through their cellular machinery. Sleep deprivation triggers changes in both how these neurons use energy and their electrical activity, suggesting that their internal state can act as a signal of sleep need. This signal is conveyed to the rest of the brain by slow, rhythmic oscillations produced by the dFBN population. As in mammals, these waves are a key marker of slumber.
As two winners of the Emerging Neuroscientist Seminar Series 2025, Drs Sarnataro and Hasenhuetl recently spoke at SWC about their work. In this article, they introduce their findings on how cellular energy balance and network-level oscillations can regulate sleep.
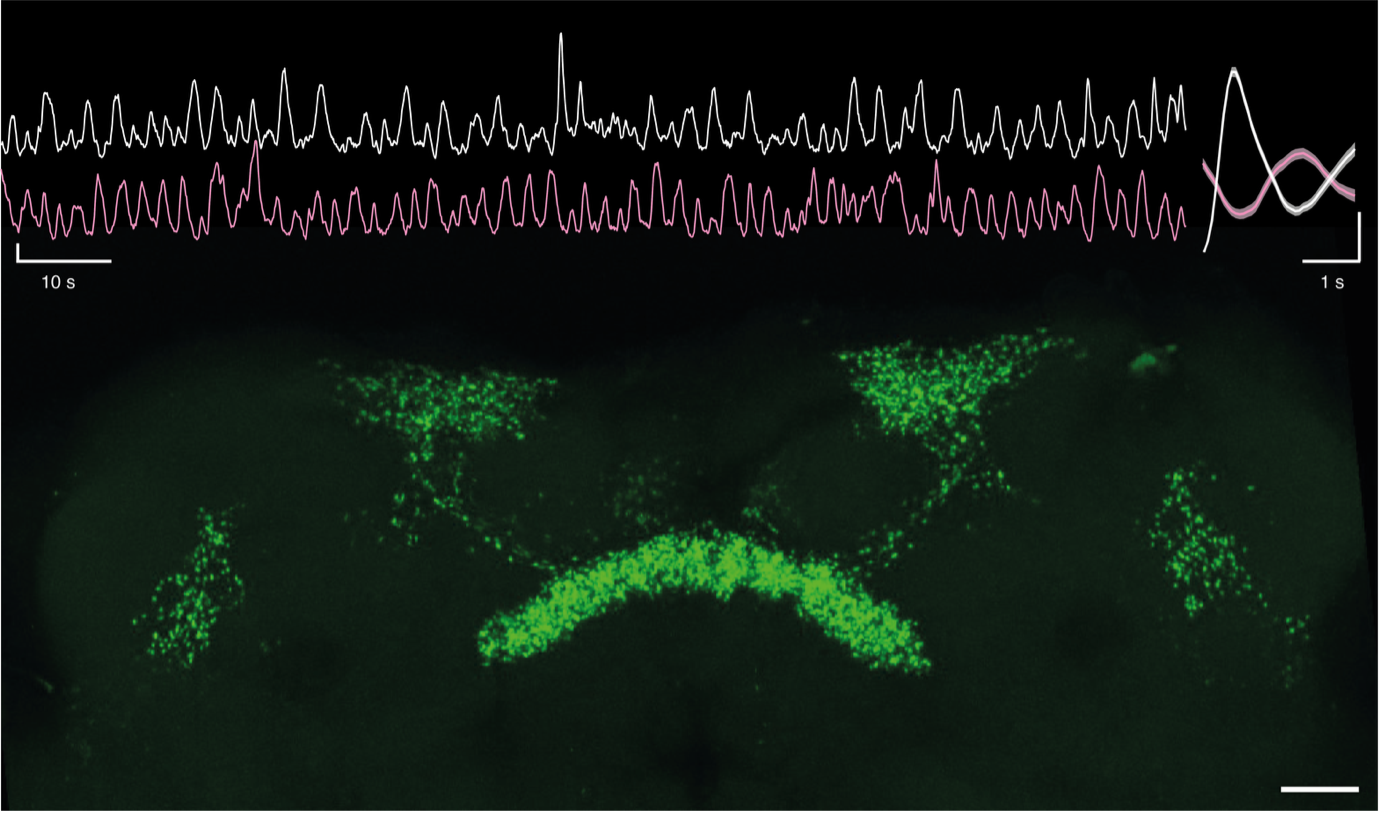
Top left, simultaneous calcium-imaging of dorsal fan-shaped body neurons in both hemispheres. Top right, average GCaMP-transient in one hemisphere (white), and the associated GCaMP-waveform on the contralateral hemisphere (red). Y-axis scale bars: 1 z-score. Bottom, maximum intensity projection of a fly midbrain where dorsal fan-shaped body neurons have their mitochondria labelled with mito-GFP. Scale bar: 20 µm.
How do you define sleep, and how much can we learn about sleep from flies?
Raffaele Sarnataro (RS): Sleep is an innate behaviour characterised by periodic, reversible quiescence accompanied by reduced responsiveness to sensory stimuli. It is such a fundamental behaviour that basic regulatory principles found in one animal can likely be extrapolated to other species.
Of course, the way sleep is defined in flies differs from how it is done in mammals. We do not have a standard electrophysiological signature of sleep in the fly. But we can monitor their behaviour. We consider a fly asleep when it is still for a few minutes. In fact, if you impair this state, flies learn poorly and become sleepier.
An extraordinary achievement of the research using fruit flies is the discovery of the core machinery of the circadian clock (recognised by the 2017 Nobel Prize in Physiology). For more than a quarter of a century, the fly has also been used as a model to study sleep, and we have discovered many commonalities between the regulation of sleep in flies and mammals.
Peter Hasenhuetl (PH): I agree with Raffaele. I want to add that it has also been quite gratifying to pay close attention to what's actually different in the ways distinct species solve problems like sleep homeostasis.
This can give important insights into the environmental and internal constraints on the respective systems. And hopefully, it will help to abstract the species-specific details from what's essential for sleep homeostasis in general.
You’ve found that slow-wave oscillations are present in the fly brain throughout the sleep–wake cycle, and their strength is a key driver of sleep. Were you surprised to find this?
PH: When we started this project, it had already been established that these dFBNs are involved in the homeostatic regulation of sleep. And there was also an elaborate dissection of some of the cell-intrinsic mechanisms that allow these neurons to sense the need for sleep. But that's not the end of the story.
We were motivated by the question of how this group of neurons produces activity patterns that other neurons can use.
We didn't expect to see this oscillatory activity using calcium imaging. We were fortunate, I think, to have chosen a recording modality with the right temporal resolution to see this.
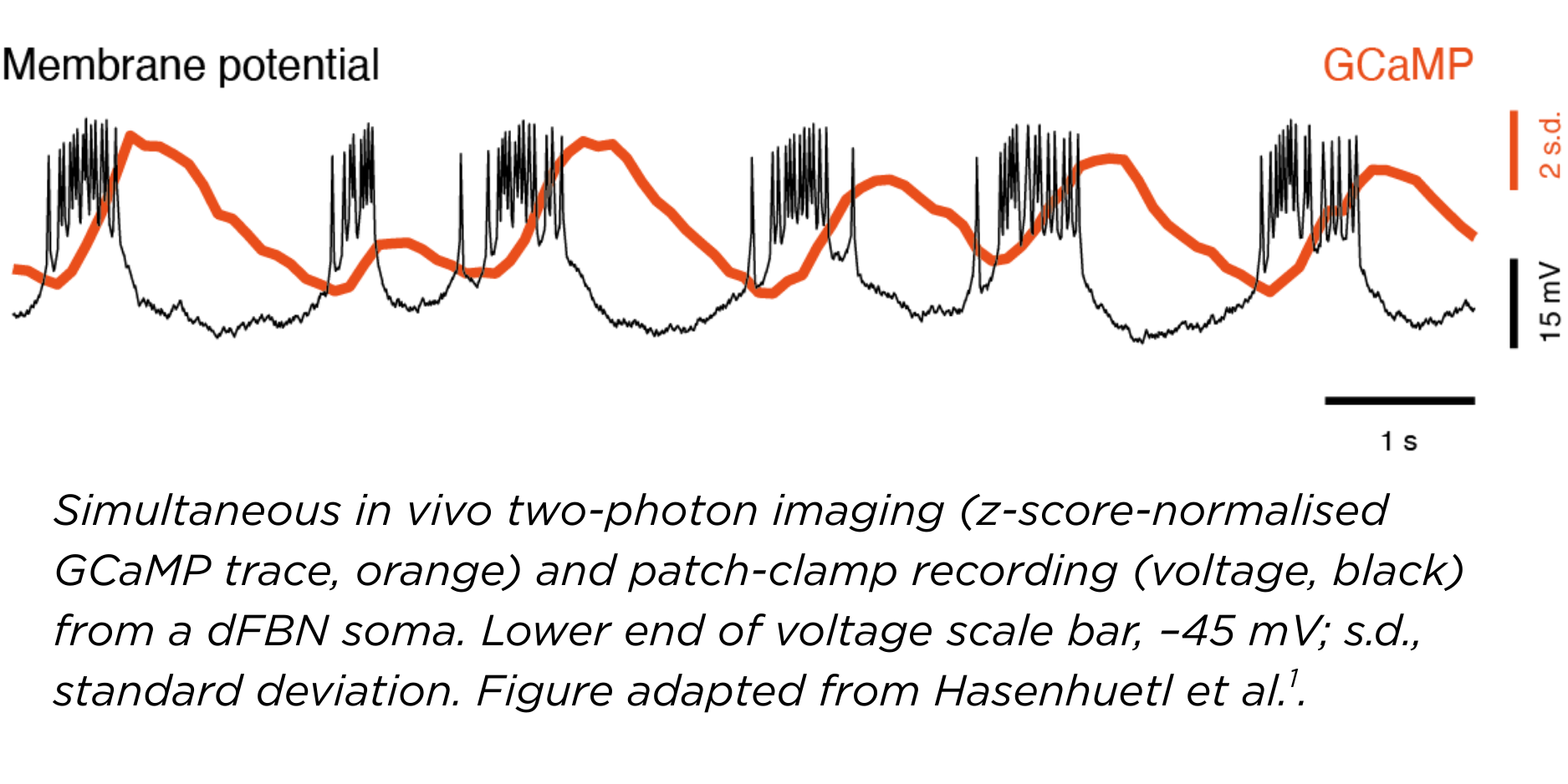
Early during this process, we were also very much encouraged by the work of another lab that found slow oscillations in a different area of the fly brain - the ellipsoid body. Both the fan-shaped body and the ellipsoid body had previously been shown to be involved in sleep homeostasis. So, we were confident we were on the right track.
It’s interesting that although the oscillations are the same frequency as those in mammals, the fly brain generates them in a different way. Their spatial spread and the persistence of these waves also differ.
We were quite surprised to see that – even though dFBNs increased their oscillatory power with sleep need and their artificial replay promoted sleep – their oscillations actually persisted in awake flies.
I think this gives first clues into how the dorsal fan body conveys sleep pressure information to downstream neurons1, which we can now begin to dissect systematically.
You have also been addressing how metabolism is affected by and affects neuronal activity. Can you tell us what you have found in relation to sleep?
RS: There were already indications that mitochondria, the cell's energy-production organelle, of sleep-control neurons, were involved in sleep regulation.
We took an unbiased approach and studied single cells across the whole brain of rested and sleep-deprived flies. We found that sleep loss specifically upregulates the expression of genes involved in cellular energy production, in particular mitochondrial respiration and ATP generation, in sleep-control dFBNs. A fortunate convergence of evidence! At the same time, we also observed that the synaptic machinery of these neurons is downregulated by the lack of sleep. This provided a first link between cell-intrinsic and network-wide signatures of sleep deprivation2.
The gene expression changes in sleep-deprived dFBNs are accompanied by structural alterations inside the cells. Mitochondria become fragmented, but this is reversible with sleep. These changes in the shape of mitochondria (fragmentation or fusion) in dFBNs altered their electrical properties and eventually their ability to control sleep.
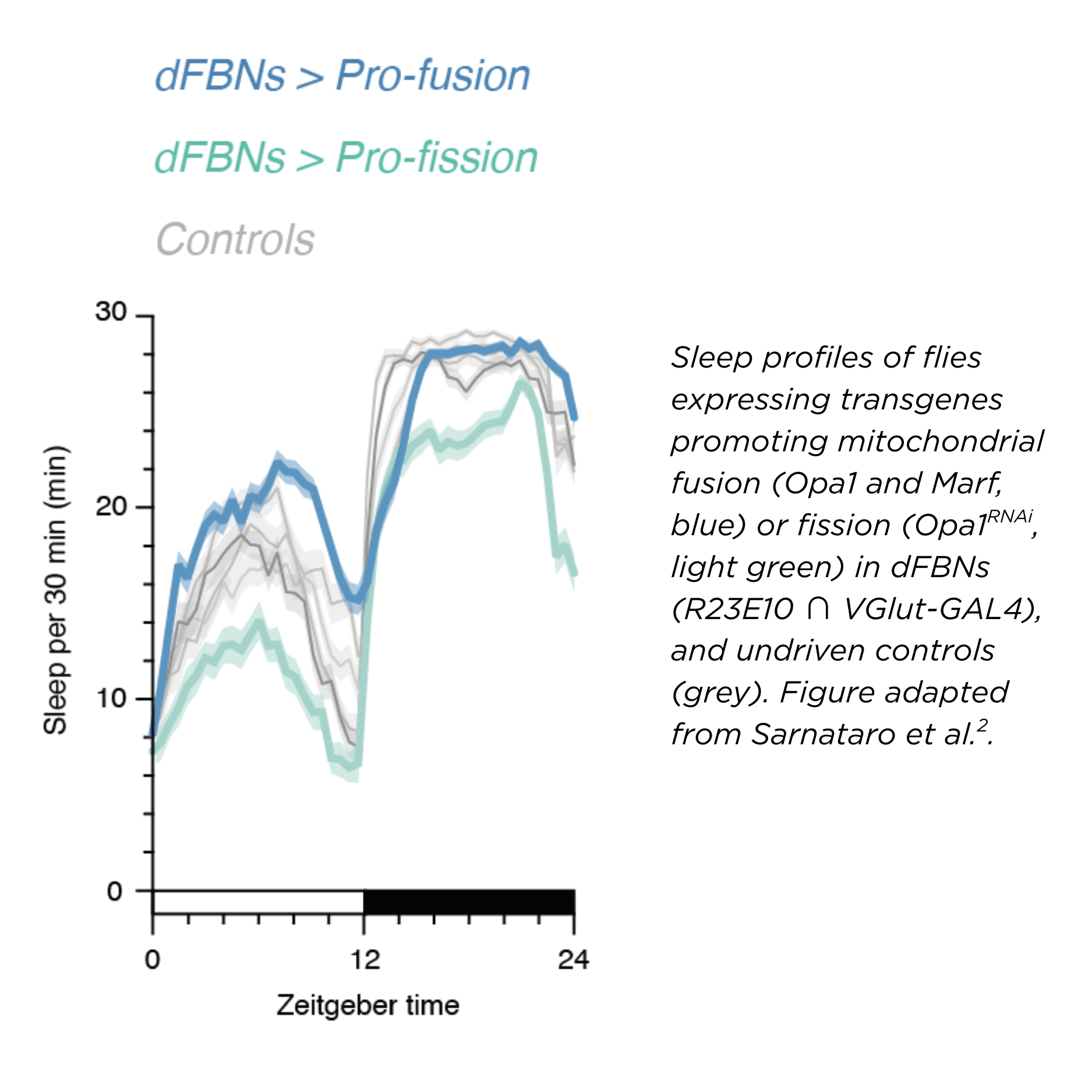
We think these shape dynamics happen as a response to an energy imbalance inside the cell – a mismatch between ATP demand and electron flow. In fact, tuning the flow of electrons, just in this small set of neurons, bidirectionally regulates a fly’s sleep.
It is known that lack of sleep poses a substantial metabolic challenge for the whole organism, but especially the brain. For its size, the mammalian brain is the body’s most energy-hungry organ, for example. Our findings suggest that sleep-control cells placed in the brain might act as sensitive early “sentinels” that detect metabolic changes. This sets them up to sense the need for sleep.
It is possible to speculate, then, that sleep evolved to fulfil a metabolic restorative need and that it is therefore an inevitable consequence of an aerobic metabolism, akin to ageing.
In your talk, you mentioned the parallels between sleep pressure in flies and hunger in mice, both mediated by mitochondria. Could you explain those links?
RS: In the mammalian hypothalamus, there is a structure called the arcuate nucleus, which includes two populations of neurons that have opposing roles in controlling feeding behaviour homeostasis.
These cells also undergo cycles of mitochondrial fission and fusion, which change the ATP levels within, and eventually their electrical activity. The resulting firing of these neurons controls the feeding behaviour of the animal.
It's fascinating that for controlling a metabolically-targeted behaviour, like feeding, cell metabolism itself is used – it's a convenient feedback loop! Interestingly, we found an analogous mechanism for sleep.
Do you think sleep is mainly a way for the brain to reset its energy balance? Or is that just part of the picture?
RS: This is a big question. What are the functions of sleep? Certainly many. Likely, a basic, ancestral, possibly biochemical necessity of neurons has driven the evolution of sleep, and over time, other functions have conveniently been crammed into this state. Disentangling them is hard.
Maybe sleep is primarily needed to clear the brain of some metabolite byproducts of neural activity, or because the connections between neurons, strengthened by the experiences of wakefulness, need to be appropriately pruned; scientists do not have a definitive answer.
Interestingly, both these hypotheses imply that extended wakefulness poses a metabolic challenge to the brain. But what is the primary factor that sleep reset? In our work, we have identified a mechanism by which sleep is controlled, potentially opening a novel window onto its core functions.
How can we leverage our understanding of the neurobiology of homeostasis to think about potential applications for metabolic disease or sleep disorders?
PH: There are many common tasks that animals with brains have to solve, and the homeostatic control of sleep is one of them. Simpler organisms actually have a long tradition in neuroscience and have been extremely useful for gaining understanding of how nervous systems solve various problems.
Following that spirit, our contribution is really to add to a mechanistic understanding of sleep homeostasis in general. But of course, we hope that this foundational understanding will consequently form a basis for also thinking about applications in the future.
What is the next big question that your work has opened up for you?
PH: Both the fan-shaped body and the ellipsoid body that I mentioned earlier are not only involved in sleep homoeostasis. They are also both crucial nuclei for navigation and context-dependent decision making. I doubt that this is a coincidence.
So the next focus for me will be the influence of an animal's internal state – so how tired or hungry it is, for example – on how it navigates through the world, or through its life if you will.
Understanding how animals are able to choose what to do next is a hard problem, in part because this process most likely involves the interplay of many distributed brain areas. That's where the compact and tractable fly brain comes in! In recent years, Drosophila neuroscience has witnessed an explosion of methodological innovations and publicly available resources, which makes this model system very powerful to do circuit neuroscience on a mechanistic level.
And with the sleep and navigation research fields having converged on these brain areas, I really think the timing is perfect to tackle this question. This will involve looking at this problem on various scales, from synaptic plasticity, neural dynamics, and the interaction of genetically-defined neural populations. So, stay tuned!
RS: For me, the big open question in the field is to reconcile the two parallel ways sleep has been looked at in the brain.
Sleep is regulated by specific neural circuits, and we are continuously learning more about the pieces of this puzzle across different animals. But at the same time, sleep changes the state of the whole brain: synaptic strengths, electrical and metabolic activity of brain cells are deeply affected by the cycling of sleep and wakefulness.
With these two concurrent phenomena, the question is, how do localised circuits orchestrate the activity of virtually the whole brain? How “tired” parts of the brain signal to these circuits that it is time to fall asleep? Do, and if so, how, those processes interact?
This is often thought to somehow occur through sleep-specific oscillations: I am also interested in what such oscillatory activity does. It is just an epiphenomenon of the sleeping brain, or does it do something fundamental to the actual cell biology of neurons?
This is a big challenge for researchers - I do not think there is an established framework for this yet.
Do you have any sleep advice for readers?
RS: Get lots of sleep!
PH: As much as you can. Especially if you are a scientist who works on sleep!

Dr Peter S. Hasenhuetl and Dr Raffaele Sarnataro.
Find out more
1. Hasenhuetl PS*, Sarnataro R*, Vrontou E, Rorsman HO, Talbot CB, Brain R, Miesenböck G (2024) A half-centre oscillator encodes sleep pressure. bioRxiv
2. Sarnataro R, Velasco CD, Monaco N, Kempf A, Miesenböck G (2025) Mitochondrial origins of the pressure to sleep. Nature 645, 722–728 (2025)


