
Understanding sensory and gastrointestinal differences in autism spectrum disorder
An interview with Professor Lauren Orefice, of Massachusetts General Hospital and Harvard Medical School, conducted by April Cashin-Garbutt
Many people with autism spectrum disorder (ASD) experience sensory abnormalities and gastrointestinal issues, but the neural circuit disruptions underlying these differences are not well understood.
In a recent SWC Seminar, Professor Lauren Orefice presented her work uncovering the role of peripheral sensory neurons, outside the brain, in somatosensory and viscerosensory processing abnormalities in ASD, and how these circuit changes contribute to additional ASD-related behaviours in mouse models for autism.
In this Q&A, Professor Orefice shares her key findings to date and the potential clinical implications for people with ASD.
What kinds of sensory abnormalities and gastrointestinal problems do people with autism spectrum disorder (ASD) experience? Do these issues typically start from a young age, or can they develop in older people with ASD?
People with ASD can experience sensory processing differences across all sensory modalities. This can include vision, audition, olfaction, gustation, and, of interest to my lab, somatosensation and viscerosensation (sensations from internal organs).
In terms of the sense of touch, symptom presentations can be complex and heterogeneous. People can experience hyper- and/or hypo-somatosensory reactivity. Tactile hypersensitivity is common, as is hyposensitivity to painful stimuli.
Hypersensitivity to light touch can pose significant challenges for autistic individuals. Difficulties with certain clothing textures, like wool, tags in clothing, haircuts, and showers are common. Social interactions can also be difficult. If you go to the grocery store or a cocktail party, there's bound to many different types of tactile inputs, including social touch.
Many autistic individuals also exhibit restricted dietary preferences that are often due to food texture issues. Gastrointestinal problems are also highly prevalent in autism, including constipation, diarrhea, bloating, and pain.
We often don't appreciate how much of our daily experiences are imbued with somatosensory experiences: how intrinsic touch is to how we experience the world, how we process it, and how we respond to it.
Why are these issues so prevalent in ASD? Are the gastrointestinal difficulties, for example, due to a typically more restricted diet or other key factors?
I think it's a combination of many factors. We hypothesize that many of these sensory-related issues are linked, and a major part of our research program aims to understand what causes these sensory differences in autism.
As I showed in the talk that I presented at SWC, we find that peripheral sensory neurons of the dorsal root ganglia that innervate the gastrointestinal tract transmit signals from the gut in an abnormal or heightened way in mouse models for autism.
There are likely other contributors to gastrointestinal problems in autism, including other cell types in the gastrointestinal tract, dietary differences, and changes in the gut microbiome. For example, a restricted diet due to heighted food texture sensitivity could impact gut microbiome diversity, nutrient absorption, motility, body weight, and gut pain. We propose that each of these components is linked and contribute to overall differences in sensory processing and physiology.

Why are the neural circuit disruptions underlying these issues not well understood? How did your prior work on the circuit organisation of somatosensation lead you to tackle this question?
The observation that there are sensory issues in autism has been documented since the 1940s, when Leo Kanner made his initial observations about autism. While autistic individuals commonly experience sensory-related difficulties, sensory processing issues were not a part of autism diagnosis until 2013, when the DSM-5 was revised.
How we diagnose autism has changed, and our approaches to thinking about autism causes and treatments have also been revised over the years. For example, treating sensory sensitivities and sensory difficulties could improve quality of life for many autistic individuals.
When we started studying somatosensory circuit function in mouse models for autism back in 2014, that was right after the DSM-5 had changed to include sensory abnormalities as part of the diagnostic criteria. One of the reasons I wanted to study this topic was because I thought it was an important scientific question, we would learn a lot about the somatosensory system development, and hoped it would reveal some important autism-related biology.
Were you surprised to find that peripheral sensory neurons (outside the brain) contribute to some of the major core symptoms of ASD in mice?
We were certainly surprised that when we found that autism risk gene mutations only the peripheral neurons could cause tactile overreactivity in mice. We observed this across multiple animal models with different genetic mutations.
We were even more surprised when we found that touch over-reactivity, due to peripheral sensory neuron changes, was sufficient to disrupt some additional autism-related behaviours, like social behaviours and anxiety-like behaviours in mice.
This is of course not meant to say that the brain is not important in autism spectrum disorder, but that neurons outside the brain, can also contribute to some autism-related phenotypes in mice. We hope this research will lead to novel and tractable therapeutic targets to improve sensory-related issues in autism, by treating the peripheral nervous system.
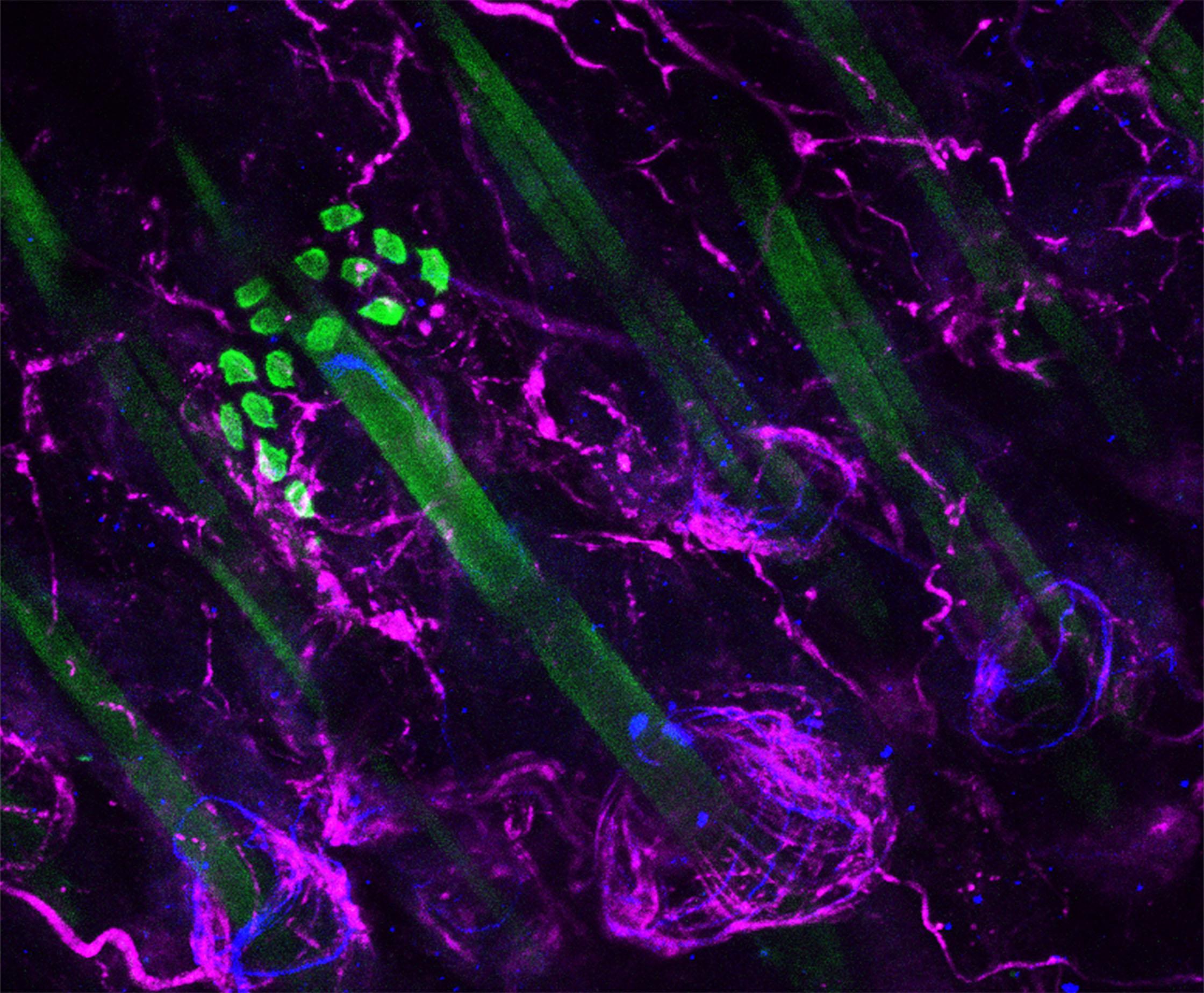
Low threshold mechanosensory neurons innervating hairy skin from a mouse. Image credit: Professor Lauren Orefice
Did you find any differences between the different mouse models for ASD?
That's a nuance I couldn't touch on in the seminar. There are many different types of autism-associated genes; some are involved in transcription regulation, others are synaptic scaffolding proteins, ion channels, or receptors, for example. These genes have a range of different biological functions in neurons and other cells of the body.
One of our approaches is to look at disparate autism-associated risk genes to understand how they might change the function of peripheral somatosensory or viscerosensory neurons and through what types of mechanisms. We find that the pathophysiology underlying dorsal root ganglion neuron dysfunction can be different across the different autism-related genes. But we find that many of these genes, regardless of the specific pathophysiology, ultimately lead to changes in how the peripheral sensory neurons detect and transmit sensory information from the cutaneous or visceral tissues.
How might altered tactile and social behaviours in mouse models inform our understanding of similar behaviours in humans with ASD?
Mice are not humans, they do not have autism, but they are a model for understanding some aspects of the very complex and heterogeneous nature of autism.
Mouse models can be quite useful for understanding specific aspects of autism biology. The sensory symptoms are something that we can model in mice, because the somatosensory and viscerosensory systems are quite similar between mice and humans. We think these are tractable circuits to try to understand using mouse models for autism.
Secondly, there are great genetic toolboxes for dissecting touch circuits and visceral circuits in mice that you can't do in a many other systems, especially in humans. This means we can manipulate gene expression patterns in a specific cell type or in a temporally precise manner; we can activate or inhibit specific cells; we can trace neurons; we can record from neurons. The power of the mouse for neuroscience research and sensory biology research is great.
Another question we can ask, for example, is how sensory processing abnormalities in mice can lead to changes in mouse social behaviours. Mice engage in a lot of social interactions, including physical social interactions, which are very complex behaviours. They are not human behaviours, but if we can understand how sensory changes can affect mouse behaviours, it might give us an inroad for understanding how sensory experiences and sensitivities may influence human behaviours, including social interactions.
What are the clinical implications of your research? Have you identified any potential therapeutic targets for addressing sensory abnormalities and gastrointestinal issues in people with ASD?
That's something we've been working on, and will continue to work on, because it’s an important and amazing opportunity for us to translate our basic biological findings into potential strategies to improve the quality of life for autistic individuals that experience sensory processing difficulties.
Our work that suggests that if you can target the functions of peripheral sensory neurons to reduce the excitability of specific types of sensory neurons and reduce the flow of sensory information from the peripheral tissues to the central nervous system, we may improve sensory processing differences.
And similarly for gastrointestinal problems, we aim to identify new ways to target the peripheral sensory neurons that innervate the colon and detect noxious stimuli, to ultimately reduce gastrointestinal pain. We know that gastrointestinal pain symptoms are tightly linked with things like irritability and social withdrawal in humans. If we can improve gastrointestinal pain, perhaps we can improve some other symptoms that are associated with it.
We are a product of our sensory worlds. Our brains don't exist in isolation. We develop by experiencing both the external and internal world, and that shapes our brains and who we are. At some level, your sensory experiences make you unique. I think that's important, that we all experience the world differently. I think it is also necessary to find ways to improve sensory processing issues, which would be beneficial for people that experience difficulties with various sensory encounters.
What are the next steps for your research?
We've become fascinated by understanding how viscerosensory circuits develop and how sensory information from our internal organs in encoded by the peripheral and central nervous systems. We want to understand viscerosensory features are encoded and represented throughout the brain, and how gastrointestinal issues may influence social and other behaviours in mouse models for autism.
We are also very interested in understanding how various sensory experiences shape brain development and modulate complex behaviours. For example, we are exploring how somatosensory experiences can shape long-range connectivity of somatosensory cortex with many other brain regions to regulate social behaviours in mice.
We're also interested understanding oral somatosensory circuits, how we detect and encode texture stimuli within the mouth, and how this information is represented in the central nervous system. We are also exploring how oral somatosensory circuits may be different in mouse models for autism.
We have lots of active projects and many wonderful lab members working on these exciting questions!
About Professor Orefice
Lauren Orefice received her Ph.D. from Georgetown University, where she studied the role of local BDNF synthesis in dendritic spine morphogenesis under the guidance of Dr. Baoji Xu. For her postdoctoral work, Lauren joined Dr. David Ginty’s Lab at Harvard Medical School and focused on understanding somatosensory alterations in mouse models for autism spectrum disorders. Her work identified that peripheral somatosensory neurons, outside the brain, are dysfunctional in multiple mouse models for ASD and contribute to the development of some of the major, core symptoms of ASD in mice. Since 2019, Lauren is an Assistant Professor in the Department of Molecular Biology at Massachusetts General Hospital and the Department of Genetics at Harvard Medical School. The Orefice lab studies the development and function of somatosensory and viscerosensory systems, with a focus on understanding how sensory circuits are altered in people with developmental disorders. Lauren has received numerous awards in recognition of her research, including the 2019 Eppendorf and Science Prize in Neurobiology, a 2020 Pew Scholar award, a 2020 Searle Scholar award, a 2021 New York Stem Cell Foundation Robertson Investigator award, a 2022 McKnight Scholar award, and a 2023 Kavli fellow award.


